Magnesium Enhanced Reactivity of High Energy Composites
Full Description
Background
Boron (B) is regarded as a premier candidate fuel in high-energy composites due to its higher reaction enthalpies. However, boron suffers from sluggish oxidation and energy release kinetics as a result of its low melting oxide shell. Post-melting, the non-volatile liquid oxide layer acts as a diffusion barrier to the oxidizing species and restricts their access to the boron core, thereby inhibiting oxidation and energy release.
Technology
Prof. Zachariah and his team have developed an innovative magnesium/boron (Mg/B) based composite that offers a thermodynamically and kinetically viable source of highly reactive gas-phase Mg that acts as an etchant for the oxide shell of boron. The developed composite creates a pristine and accessible fuel surface for the reaction. Further reactions are facilitated by the thinning of the oxide shell. Together, these serve to enhance the combustion of boron.
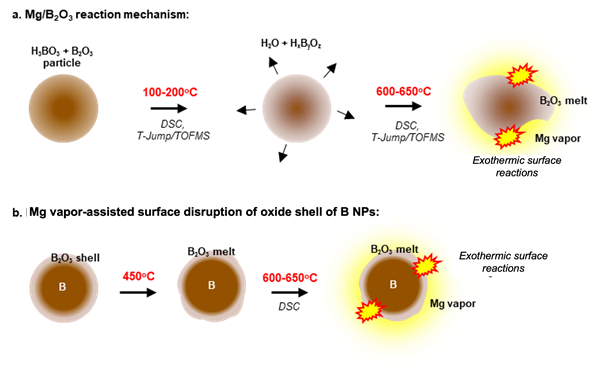
Schematic illustration of the reactivity enhancement of Boron
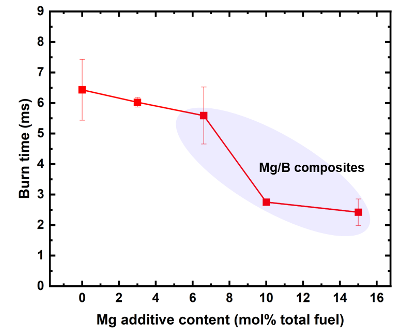
Graph showing the burn characteristics of B/CuO nanoenergetic composites. Mg/B/CuO composites show significant enhancement in burn times and pressurization over B/CuO composites.
Advantages
The significant benefits and aspects of this Mg/B-based composites relative to B-based composites are:
- A 6-fold enhancement in pressurization rates.
- A 30% increase in peak pressures.
- Shortened burn time - from 6.5 milliseconds (ms) to ~2 ms - a 60% reduction.
- Synergistic effect of the Mg and B fuels responsible for the augmentation of reactivity.
- Potential for similar augmentation for high-energy composites based on nanoscale metals and metalloids such as Aluminum, Titanium, Silicon, etc.
Suggested uses
Applications that use energetic materials such as propellants, solid fuels, thermites, etc.
User Defined 1
- Please review all inventions by Prof. Zachariah and his team at UCR.
- Please read recent press coverage of Prof. Zachariah, at UCR.
- Please visit Prof. Zachariah's research group website to learn more about their research.
Related Materials
Patent Status
| Country | Type | Number | Dated | Case |
| United States Of America | Published Application | 20240190789 | 06/24/2024 | 2023-971 |
Contact
- Venkata S. Krishnamurty
- venkata.krishnamurty@ucr.edu
- tel: View Phone Number.
Other Information
Keywords
energetic composites, thermites, propellants, pyrotechnics, solid fuels
