Safety Scalpel for Removal of Gastrostomy Tubes
Abstract
A researcher at the University of California, Davis has developed a safety scalpel for mitigating the challenges associated with the removal of certain types of percutaneous implants, such as gastrostomy tubes having non-reducible anchoring elements.
Full Description
A percutaneous implant is an object that is placed through the skin and other tissue to create a permanent or semi-permanent pathway or port through the skin into the body. An example of a percutaneous implant is a gastrostomy tube. There are various types of gastrostomy tubes including, for example, percutaneous endoscopic gastrostomy (PEG) tubes, radiologically inserted gastrostomy (RIG) tubes, and surgically placed gastrostomy tubes. Generally, a gastrostomy tube is a device placed through the abdominal wall into the stomach of a patient to allow food, fluid, drugs, and/or other substances to be introduced or administered directly into the stomach, or to allow ventilation and drainage in the case of a downstream bowel obstruction. Gastrostomy tubes are indicated when, for example, ingestion via the mouth is insufficient or impossible and the time to recovery of per oral feeding is expected to be too long for nasoenteric tubes.
A gastrostomy tube typically has an anchoring element that is positioned within the stomach to prevent inadvertent removal. The anchoring element may be comprised of a radially extending flange that can be difficult, or even traumatic, to reduce. One method of removing a gastrostomy tube, often referred to as the “cut and push” method, involves severing a portion of the gastrostomy tube outside of the body and pushing a remainder of the gastrostomy tube into the stomach, in the hopes that this portion of the gastrostomy tube, which includes the anchoring element, passes naturally through the patient’s gastrointestinal (GI) tract. This method involves a risk that the gastrostomy tube does not pass naturally and creates an obstruction in and/or perforates the GI tract and/or has other undesirable effects. To mitigate such complications, in certain cases an endoscopic or surgical procedure will be performed to retrieve the retained portion of the gastrostomy tube. Such procedures are invasive and for frail patients may not be possible due to the risk of exacerbating other medical conditions. However, the conventional method for removal is traction removal, per the device instructions for use. This conventional removal method is associated with tearing along the stoma tract, pain, bleeding, and site infection.
Accordingly, there is a need for improved tools and methods for removing percutaneous implants such as gastrostomy tubes. In response to this need, a researcher at the University of California, Davis has developed a novel device that mitigates the aforementioned challenges, especially with respect to the removal of mushroom-type gastrostomy tubes. In particular, the device allows for safe fractionation of the tube without embolization of the fractionated pieces.
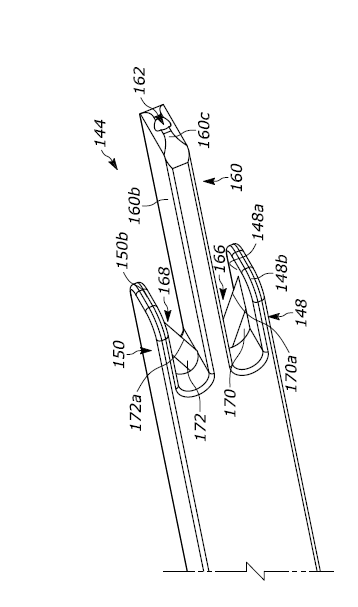
The device is configured in such a way that permits blade(s) to cute the tube without injuring adjacent tissue. Either sharp or blunt blades (e.g.., to provide an alternate method of energy delivery) may be used. The device may also be used in conjunction with a guidewire for passage of the device into a stoma tract. The device includes a proximal end having a handle and a distal end having two or three arms, with one or two cutting elements positioned between the arms. At least a portion of one of the arms extends beyond the cutting element(s) to prevent or inhibit the cutting element(s) from cutting tissue that is adjacent to the percutaneous implant when the device is used to cut the percutaneous implant. An example of the device is shown below.
Applications
- Removal of percutaneous implants such as mushroom-type gastrostomy tubes.
Features/Benefits
- Reduced incidence of complications (e.g., bleeding, tissue trauma) associated with gastrostomy tube removal.
- Reduced procedure time for gastrostomy tube removal.
Patent Status
| Country | Type | Number | Dated | Case |
| United States Of America | Published Application | 20240374278 | 11/14/2024 | 2021-621 |
Other Information
Contact
- Byron N. Roberts
- bnroberts@ucdavis.edu
- tel: View Phone Number.
Inventors
- Goldman, Roger
Other Information
Keywords
medical device, safety scalpel, implant, percutaneous, gastrostomy, surgery
