Phosphorus Pentoxide Additive for Lithium-ion Batteries
Patent Status
| Country | Type | Number | Dated | Case |
| Patent Cooperation Treaty | Published Application | WO 2023/091715 | 05/25/2023 | 2022-856 |
Additional Patents Pending
Full Description
Background
Chemical and electrochemical instability between lithium (Li) metal anode and the liquid Li-ion electrolytes result in mossy Li deposit causing severe volume expansion, low Li deposition-stripping efficieency, formation of dead Li and fast depletion of electrolyte. All of these lead to short cycle life and safety concerns. Current strategies to implement Li metal anode use solid-state Li-ion electrolytes including inorganic or organic Li-ion conductors. The disadvantages of solid-state electrolytes include poor producibility, poor stability and low conductivity. The other strategy to implement Li metal anode is to use a new type of liquid electrolytes named localized high concentration electrolytes (LHCEs). The disadvantage of the LHCEs is the expensive co-solvents which are also not readily available.
Technology
Prof. Juchen Guo and his research team at UCR have discovered the use of phosphorous pentoxide (P2O5) as an additive to modify the conventional carbonate solvents-based lithium hexa-fluoro-phosphate (LiPF6) electrolytes. P2O5 as an additive, stabilizes the electrolyte/electrode interphases by mitigating the hydrogen fluoride (HF) induced side reactions. The P2O5 additive not only improves th Li metal deposition morphology for a denser deposition layer but also alleviates the transition metal dissolution and NMC622 particle cracking problems.
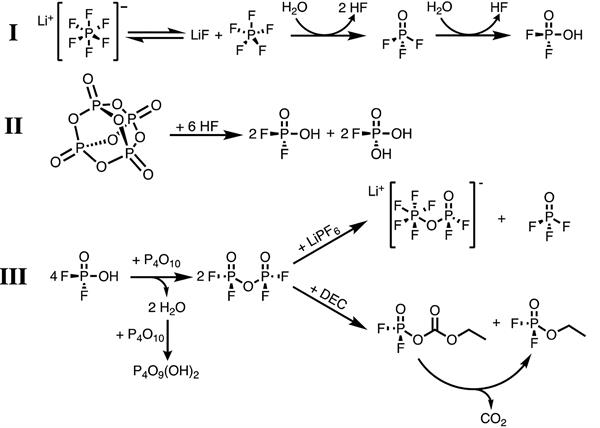
Mechanisms of P2O5 additive: (I) Decomposition of LiPF6 electrolyte induced by H2O to produce HF and difluorophosphoric acid (HPO2F2); (II) HF scavenging reaction of P2O5; (III) Reactions of products from (II) with P2O5 to generate new species beneficial to the Li metal anode.
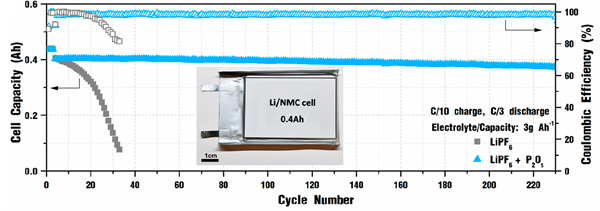
Cycling performance of 0.4 Ah Li||NMC622 pouch cell (50 µm Li anode and 3 mAh/cm2 cathode areal capacity) in lean electrolyte (electrolyte to capacity ratio = 3 g per Ah) at C/10 charging and C/3 discharging.
Advantages
- Mitigation of lithium metal corrosion.
- Greatly improved cell performance because of mitigation of HF induced side reactions and improved Li metal deposition.
- Improved cycle life - the cycle life of a 0.4 Ah Li-NMC622 pouch cell improved from 30 cycles to more than 200 cycles with 87.7% capacity retention.
- Li-NMC622 cathode resistance stays relatively stable.
Suggested uses
Rechargeable lithium-ion batteries
State Of Development
Inventors have developed and demonstrated a proof-of-concept, rechargeable Li-metal batteries using this new electrolyte.
Inventor Information
- Please visit Prof. Guo's research group website to learn more about their research.
- Please review all inventions by Prof. Guo and his team at UCR.
Related Materials
Contact
- Venkata S. Krishnamurty
- venkata.krishnamurty@ucr.edu
- tel: View Phone Number.
Other Information
Keywords
lithium ion battery, LiPF6, electrolyte, Li metal anode, lithium metal battery, NMC622, cycle life
